When two or more drugs are combined into a single pill or formulation, getting the dose right isn’t just about matching numbers-it’s about making sure the body responds the same way every time. This is the core challenge of therapeutic equivalence in combination products. Patients rely on these combinations for conditions like high blood pressure, diabetes, depression, and high cholesterol. But when a pharmacist swaps a brand-name combo for a generic, or one generic for another, subtle differences in how the drugs are made can change how well they work-or even cause side effects.
What Does Therapeutic Equivalence Really Mean?
Therapeutic equivalence means two drug products can be swapped without changing the patient’s outcome. The U.S. Food and Drug Administration (FDA) calls this the "A" rating in the Orange Book, a public database that lists approved drug products and their equivalence status. To earn that rating, the products must have the same active ingredients, in the same amounts, in the same form (like tablets or capsules), and given the same way (by mouth, injection, etc.).It’s not enough that both pills contain, say, amlodipine and benazepril. They must contain exactly 5 mg of amlodipine and 10 mg of benazepril. If one has 2.5 mg and the other has 5 mg, they’re not equivalent-even if they’re from the same company. The FDA tracks over 14,000 drug products with therapeutic equivalence ratings, and about 95% of them are rated "A." That means most generics are safe to swap.
But here’s where it gets tricky: combination products. When two drugs are mixed, their interaction isn’t always simple. One drug might boost the effect of the other, or change how fast it’s absorbed. For example, tramadol and acetaminophen together work better than either one alone. That synergy means you can’t just assume that if 50 mg of tramadol + 325 mg of acetaminophen is equivalent to another brand, then 75 mg + 325 mg will behave the same. Dose equivalence becomes a math problem, not just a label check.
Why Different Doses in Combinations Cause Problems
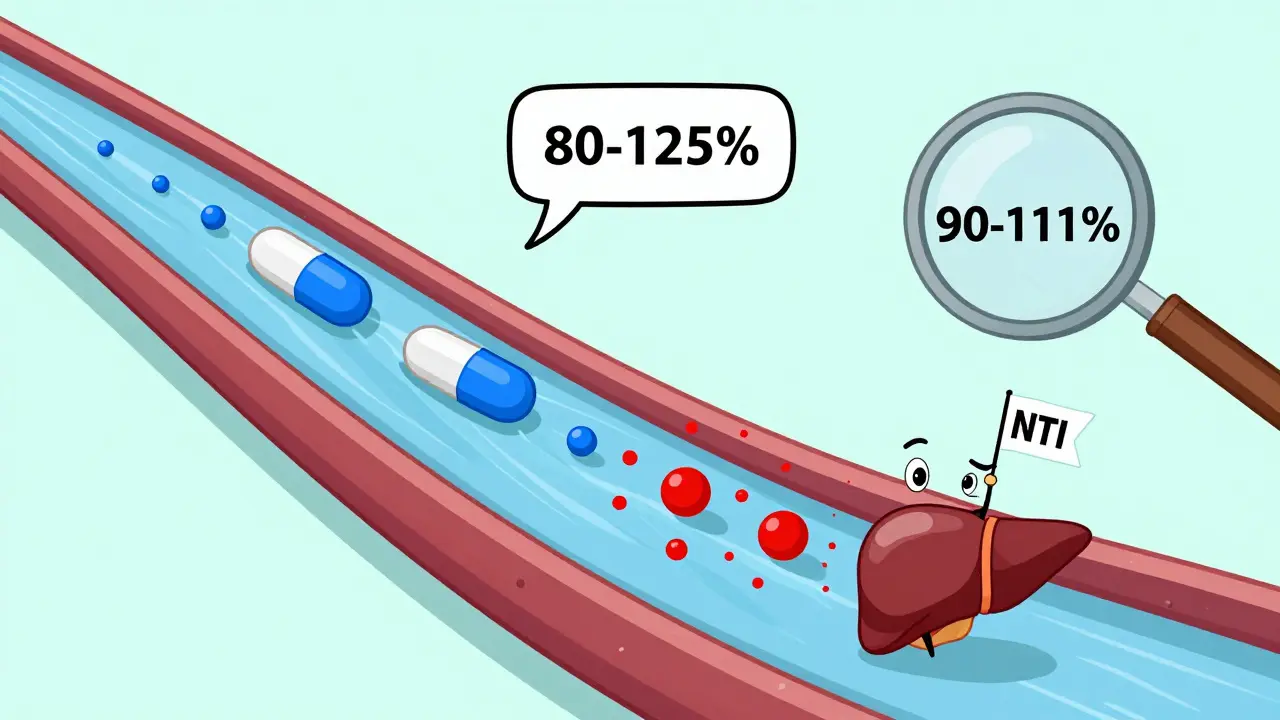
Not all combination products are created equal-even if they have the same name. Take the cholesterol drug Vytorin (ezetimibe/simvastatin). When a patient switched from the brand to a generic, their LDL (bad cholesterol) rose by 15%. Why? The generic had the same active ingredients, same doses, same form. But the inactive ingredients-fillers, binders, coatings-were different. These might seem harmless, but they can affect how fast the drugs dissolve in the gut. In some patients, that small delay meant less drug got into the bloodstream.That’s why the FDA requires bioequivalence studies: they measure how much of the drug actually gets into the blood and how fast. For single drugs, the acceptable range is 80% to 125% of the brand’s levels. But for drugs with a narrow therapeutic index (NTI)-where the difference between a helpful dose and a toxic one is tiny-this range tightens to 90% to 111%. Drugs like warfarin, levothyroxine, and phenytoin fall into this category. When they’re part of a combo, the risk of error multiplies.
A 2018 study found that 12% of patients switching between different brands of levothyroxine (even though they were all rated "A") had symptoms like fatigue, weight gain, or heart palpitations. These weren’t random mistakes. They were caused by tiny differences in how the body absorbed the hormone. And when levothyroxine is combined with another drug-say, a beta-blocker for heart rhythm-the impact can be harder to spot.
How the FDA Rates Combination Products
The FDA uses a letter-code system to rate therapeutic equivalence. "A" means the product is interchangeable. "B" means there’s uncertainty-maybe the bioequivalence data is incomplete, or the formulation differs in a way that could matter.For combination products, the rating depends on how the product was approved. Most generics come through an Abbreviated New Drug Application (ANDA). These usually get "A" ratings if they match the brand exactly. But some combos are approved under a 505(b)(2) pathway, which allows changes to the original drug-like a new coating or a different release mechanism. These may get "A" or "B" ratings depending on whether those changes affect how the drugs work.
Take the asthma combo Advair Diskus (fluticasone/salmeterol). When the generic version came out, it had the same active ingredients, same delivery device, and same doses. It was rated "A" and cost 40% less. But another combo, say, amlodipine/valsartan, might have five different generic versions, each with a different filler. Three use croscarmellose sodium as a disintegrant. Two use sodium starch glycolate. Both are approved. Both are "A" rated. But in patients with slow gut motility, one might absorb slower than the other. That’s not a flaw-it’s a reality.
Real-World Mistakes and How to Avoid Them
Pharmacists and doctors report consistent problems with combination products. One pharmacist on Reddit said they made three dosing errors in six months just from switching between different strengths of amlodipine/benazepril combos. Why? Because the pills looked similar, but the dosing wasn’t labeled clearly. Another nurse practitioner saw a patient’s cholesterol spike after switching generics-because the new version had a different coating that delayed absorption.The FDA’s Adverse Event Reporting System recorded 247 incidents in 2022 related to dose errors in combination products. Thirty-seven percent involved heart medications. Twenty-nine percent involved psychiatric drugs like sertraline/olanzapine or fluoxetine/bupropion. These aren’t rare. They’re predictable.
Here’s how to reduce risk:
- Always check the TE code in the FDA Orange Book before substituting. Don’t rely on drug names alone.
- Use barcode scanning for all combination products. It catches mismatches in strength and manufacturer.
- Don’t assume equivalence across strengths. A 5/10 combo isn’t the same as a 10/20 combo just because the numbers doubled. The ratios matter.
- Monitor high-risk patients for 72 hours after switching. Especially if they’re on NTI drugs like warfarin, lithium, or levothyroxine.
- Document substitutions. If you switch a patient from Brand X to Generic Y, note it in their chart. If they have a reaction, you’ll need that trail.
What’s Changing in 2026?
The FDA is moving toward smarter tools. In 2023, they released draft guidance for complex combination products, recognizing that dose-response curves aren’t always linear. One drug might work better at low doses but plateau at higher ones. Another might become toxic faster. Simple math won’t cut it anymore.They’re also testing machine learning models that predict which generic substitutions might fail based on formulation differences. Early tests got 89% accuracy. That’s not perfect-but it’s a start.
Looking ahead, personalized medicine could change everything. By 2030, the NIH expects 30% of therapeutic equivalence decisions to include genetic data. For example, if a patient metabolizes drugs slowly due to a CYP2D6 gene variant, a combo that works for most people might be too strong for them. The "A" rating might still apply-but the doctor might choose a different product anyway.

Bottom Line: Equivalence Isn’t Always Equal
Therapeutic equivalence is a powerful tool. It saves the U.S. healthcare system over $1.7 trillion a decade. Generic combos make treatment affordable. But equivalence doesn’t mean identical. It means "likely to behave the same in most people."For patients on combination drugs, especially those with chronic conditions or narrow therapeutic windows, the difference between "A" and "A" can be real. Pharmacists need to be vigilant. Doctors need to ask. Patients need to speak up.
When you’re managing a combo, don’t just check the label. Check the TE code. Check the manufacturer. Check the patient’s history. Because when it comes to dose and combination, the smallest change can have the biggest impact.
Can I switch between different generic versions of a combination drug?
Yes-if both versions have the same FDA therapeutic equivalence (TE) code (usually "A"). But switching between generics with different inactive ingredients can sometimes affect absorption, especially for drugs with a narrow therapeutic index. Always monitor patients closely after switching, and avoid multiple switches unless necessary.
Are all generic combination drugs as effective as brand-name versions?
Most are, but not all. The FDA requires generics to meet strict bioequivalence standards. However, some patients report differences in side effects or effectiveness, often due to variations in inactive ingredients or manufacturing processes. A 2018 study found 12% of patients on levothyroxine combos had symptoms after switching generics, even though all met FDA standards.
What is a narrow therapeutic index (NTI) drug, and why does it matter in combinations?
An NTI drug has a very small window between an effective dose and a toxic one-examples include warfarin, levothyroxine, and phenytoin. In combinations, even tiny differences in absorption can push a patient into toxicity or underdosing. The FDA requires stricter bioequivalence testing (90-111% range) for NTI drugs compared to the standard 80-125% range.
Why do some combination drugs get an "A" rating while others get a "B"?
An "A" rating means the product is therapeutically equivalent to the brand-name version. A "B" rating means there are unresolved bioequivalence issues-perhaps because the formulation differs in a way that could affect absorption, or because the data is incomplete. Combination products approved under the 505(b)(2) pathway are more likely to get "B" ratings if they include changes like new coatings or release mechanisms.
How can I find the therapeutic equivalence rating for a combination drug?
Use the FDA’s Orange Book, available online at the FDA’s website. Search by brand name or active ingredients. Look for the TE code-"A" means interchangeable, "B" means not recommended for substitution. Pharmacists should check this before dispensing or substituting any combination product.
Do drug interactions affect therapeutic equivalence in combinations?
Yes. Therapeutic equivalence assumes each drug behaves the same way in isolation. But in combinations, drug-drug interactions can change how one component is absorbed or metabolized. For example, a CYP3A4 inhibitor might increase the level of one drug in a combo, making the whole product behave differently than expected. This isn’t captured in standard bioequivalence studies and requires clinical judgment.
Next Steps for Clinicians and Patients
If you’re a clinician: Always verify the TE code before approving a substitution. Document any switches in the patient record. Consider using a standardized conversion table for NTI drug combos. If a patient reports a change in symptoms after switching, don’t dismiss it-check levels if possible.If you’re a patient: Ask your pharmacist if the drug you’re getting is a generic, and whether it’s the same as your previous version. If you’re on a combo with a narrow therapeutic index, ask if the manufacturer changed. Keep a list of your medications-including strengths and brands-so you can spot changes.
Therapeutic equivalence is meant to make care safer and cheaper. But it’s not foolproof. The best system is one that’s backed by data, monitored closely, and respected by everyone involved.


Post A Comment